When the ponds where one
African fish lives dry up, its offspring put their lives on pause. And now
researchers have a sense for how the creatures do it.
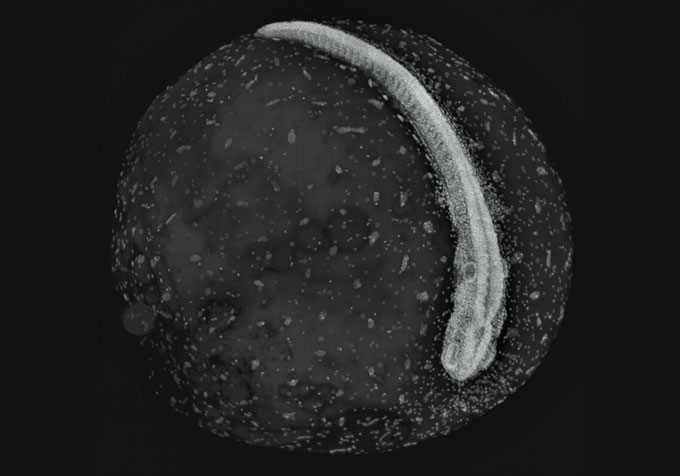
African turquoise killifish embryos
can halt their development during
a state of suspended activity called diapause. Now a study shows that the embryos
effectively don’t age while in that state. Genetic analyses reveal that, to
stay frozen in time, the embryos put functions
such as cell growth and organ development on hold, researchers report in the Feb. 21 Science.
“Nature has identified ways
to pause the clock,” says Anne Brunet, a geneticist Stanford University.
Knowing how killifish pause their lives could help scientists figure out how to
treat aging-related diseases or learn how to preserve human organs long-term,
she says.
Nematode worm larvae (Caenorhabditis elegans) can also halt
development and aging when faced with a lack of food or if their environment is
overcrowded. Invertebrates
like nematodes, however, lack many of the features that make other animals age,
such as an adaptive immune system. More
than 130 species of mammals from mice to bears also have some form of diapause.
The killifish (Nothobranchius furzeri) live in ponds in Mozambique and Zimbabwe that disappear for months during the dry season, leaving the fish without a home until the rain returns (SN: 8/6/18). For adults that typically live only four to six months anyway, vanishing ponds don’t pose much of a threat. But some killifish embryos press pause on their development during dry months, until ponds fill up again.

Killifish
embryos can put their growth on hold from five months up to two years, matching
or even greatly exceeding their typical adult life span. If humans could do
something similar, an 80-year-old person might instead have a life span from 160
to more than 400 years, Brunet says. But if, or how, these animals protect
themselves from aging while in this limbo was unknown.
In the study, Brunet and her
colleagues compared killifish embryos that halted their growth with those that
bypassed diapause and hatched into adults. Diapause didn’t decrease an adult
fish’s growth, life span or ability to reproduce — a sign that the animal didn’t age, even if it paused its
development for longer than its typical lifetime, the researchers found.
The team
then analyzed the genetic blueprint of embryos suspended in diapause to
determine which genes were active. Although the young killifish had developing
muscles, hearts and brains before diapause, genes involved in organ development
and cell proliferation were subsequently turned off. But other genes were
cranked up, such as some crucial for turning other sets of genes on or off.
One gene,
the chromobox 7 gene, or CBX7,
repressed genes involved in metabolism, but turned on those important for
maintaining muscle and staying in diapause, the researchers found. Embryos
without CBX7 came out of diapause
sooner, and their muscles began to deteriorate after one month.
The new
study shows that the embryos aren’t passively waiting for better environmental
conditions — their cells coordinate responses during diapause that protect
killifish from the passage of time. “We have
always looked at this diapause state as more passive — nothing happens there,” says Christoph Englert, a molecular geneticist
at the Leibniz Institute on Aging in Jena, Germany, who wasn’t involved in the
work. But the new research “shifts the paradigm of diapause as a passive,
boring state to an active state of embryonic nondevelopment.”
Researchers aren’t sure how things
like temperature might spark a developing killifish to begin or end diapause. But
understanding what’s going on inside an embryo is a step toward pinpointing how
external signals might control when the animals suspend time, Englert
says.
